Introduction to Computational Chemistry - Third Edition
Frank Jensen
Department of Chemistry, Aarhus University, Denmark
Introduction
Chemistry is the science dealing with construction, transformation and properties of molecules.
Overview
“many-body problem”
Systems composed of more than two particles cannot be solved by analytical methods, but Computational methods.
the Breit term
$$ \begin{equation} \mathbf{V}_{\text {elec }}\left(\mathbf{r}_{12}\right)=\frac{1}{r_{12}}\left[1-\frac{1}{2}\left(\boldsymbol{\alpha}_{1} \cdot \boldsymbol{\alpha}_{2}+\frac{\left(\boldsymbol{\alpha}_{1} \cdot \mathbf{r}_{12}\right)\left(\boldsymbol{\alpha}_{2} \cdot \mathbf{r}_{12}\right)}{r_{12}^{2}}\right)\right] \end{equation} $$
The first-order correction is known as the Breit term, and α1 and α2 represent velocity operators. The first term in Equation (1.4) can be considered as a magnetic interaction between two electrons, but the whole Breit correction describes a “retardation” effect, since the interaction between distant particles is “delayed” relative to interactions between close particles, owing to the finite value of c (in atomic units, c ∼ 137).
Dimains of dynamical equations
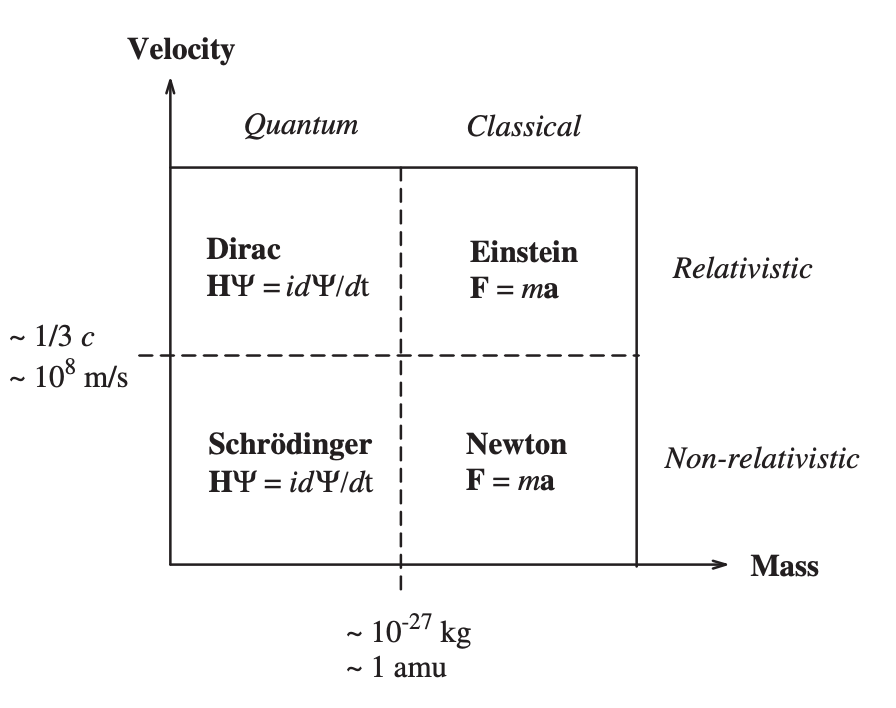
potential energy surface (PES)
one of the major problems is calculating the electronic energy for a given nuclear configuration to give a potential energy surface.
Force Field Methods
also referred to as molecular mechanics (MM) methods.

